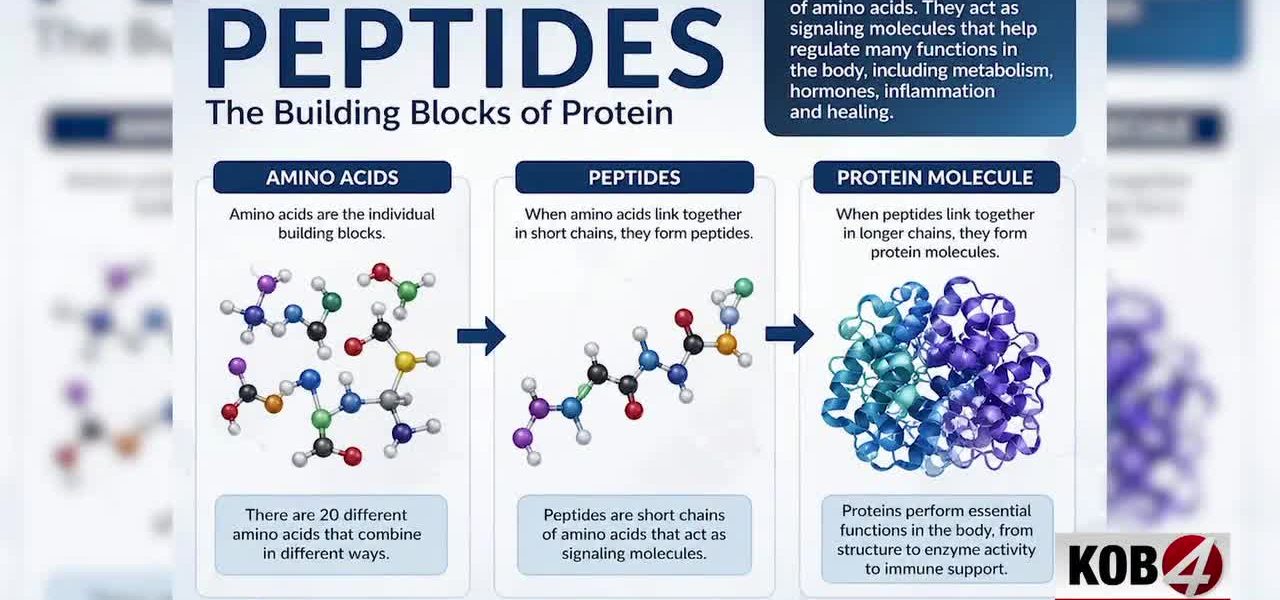
The Peptide Paradox: Unregulated ‘Youth Serums’ Spark Policy Headaches, Consumer Peril
POLICY WIRE — ALBUQUERQUE, N.M. — The fountain of youth, it turns out, might just be a vial of synthetic amino acids, dispensed by an aesthetician and promising everything from svelte waistlines to...
POLICY WIRE — ALBUQUERQUE, N.M. — The fountain of youth, it turns out, might just be a vial of synthetic amino acids, dispensed by an aesthetician and promising everything from svelte waistlines to wrinkle-free complexions. Never mind the meticulous scientific vetting, the rigorous clinical trials, or the watchful eye of federal regulators; the burgeoning peptide market, peddled across storefronts and digital channels, has largely sidestepped such bureaucratic inconveniences.
It’s a peculiar brand of modern alchemy flourishing in a regulatory grey zone, from sun-baked New Mexico clinics to the sprawling metropolises of South Asia. Consumers, eager for a shortcut to wellness or an elusive grasp on fading youth, are flocking to these treatments, often with little understanding of their long-term efficacy or potential risks. At its core, this isn’t just about vanity; it’s a rapidly expanding frontier of public health policy — or the stark absence of it.
The substances in question, peptides, are short chains of amino acids, the building blocks of proteins. They’re naturally occurring — and vital for countless biological processes. Insulin, for instance, is a peptide hormone. But the peptides now flooding the wellness market are often synthetic concoctions, marketed with vague claims of boosting collagen, accelerating metabolism, or enhancing muscle growth. The problem, for policy architects, isn’t their existence but their classification: are they drugs, cosmetics, or dietary supplements?
“The regulatory framework simply hasn’t caught up to the explosive growth in these quasi-pharmaceutical compounds,” shot back Dr. Evelyn Reed, Deputy Director of the U.S. Food — and Drug Administration’s (FDA) Office of Non-Prescription Drugs, in a rare candid moment. “When products make therapeutic claims—altering body functions for disease treatment or prevention—they typically fall under drug regulations. But many of these peptide purveyors dance around that definition, presenting them as ‘cosmetic’ or ‘research chemicals.’ It’s a legal tightrope act, perilous for consumers.”
And peril, as it happens, isn’t always obvious. Adverse reactions range from mild skin irritation to more serious systemic issues, yet reporting mechanisms remain fractured and inconsistent. Still, the global peptide therapeutics market was valued at an astounding USD 49.3 billion in 2022 and is projected by Grand View Research to reach USD 75.3 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 9.2%. That’s a lot of potential profit—and a lot of potential problems—being brewed in an opaque industry.
Behind the headlines of miraculous transformations, a more sobering reality emerges. In Albuquerque, a local aesthetician, Anna Clark, has become a de facto explainer for curious clientele, fielding questions about the latest peptide fads. She’s providing information, yes, but the broader environment lacks the robust, independent scientific scrutiny that true medical treatments demand. It’s a Wild West scenario, albeit one where the gold rush is paved with injectable youth.
This regulatory lacuna isn’t confined to American shores. In Pakistan, for instance, where beauty standards and the pursuit of youthful appearances are often intense (and heavily influenced by global trends), unregulated peptide clinics have begun to proliferate in urban centers. Without robust local oversight, consumers there might face even greater risks, compounded by less stringent import controls and potentially counterfeit products. It’s a global phenomenon, really, mirroring the free flow of information – and misinformation – that defines our interconnected age. A culture of immediate gratification clashes with the painstaking process of scientific validation.
Dr. Anya Sharma, President of the Global Anti-Aging Consortium, offered a more sanguine view, almost a lament. “Innovation often outpaces legislation; we’re providing what consumers demand, with safety protocols in place,” she asserted, a hint of exasperation in her voice. “The onus is also on consumers to research — and choose reputable providers. We don’t want to stifle progress with overly burdensome regulations, but clarity is undeniably needed.” (A clarity that, for now, remains elusive.)
So, as the global wellness industry continues its inexorable expansion, driven by aging populations and aspirational marketing, the peptide conundrum will only intensify. Policymakers, from Washington to Islamabad, are grappling with the same fundamental question: how do you regulate a market that exists largely in the shadows, promising the moon but delivering, at times, something far more terrestrial – and unsettling? The solution won’t be simple; it’s a complex weave of consumer education, legislative agility, and perhaps, a touch of legal fortitude, especially where free speech intersects with marketing claims.
What This Means
The unchecked proliferation of peptide-based ‘wellness’ products signals a profound challenge to established public health and consumer protection frameworks. Politically, this creates a fertile ground for consumer advocacy groups to demand stronger federal oversight, potentially leading to increased pressure on the FDA or similar international bodies to redefine and regulate these substances more aggressively. Economic implications are significant: a crackdown could devastate an already multi-billion-dollar industry that thrives on its current ambiguous status, but conversely, a clear regulatory pathway could legitimize it, drawing in more traditional pharmaceutical investment. For consumers, the current landscape means navigating a minefield of unsubstantiated claims and potential health risks, making informed choices exceedingly difficult. Ultimately, the peptide paradox highlights a growing chasm between rapid biotechnological innovation and the slower, more deliberate pace of governance, posing a long-term question about how societies manage technologies that blur the lines between medicine, cosmetics, and lifestyle enhancement. There’s no easy answer here, folks.